Taking into account the biological differences of patients
Precision medicine’s objective is “to optimize a specific preventive, diagnostic or therapeutic intervention in a given subpopulation of patients, which would most likely to benefit from it” (citation from “California Initiative to Advance Precision Medicine”).
The development of Precision Medicine is based on the use of large numbers of data (Big data) and constitutes a considerable transformation of medical practices and underlying concepts. Providing predictive values on the quality and the efficiency of a treatment will significantly improve the patient medical care and its quality of life during treatment, but also reduce hospitalization costs, expenses in biomedical research and pharmaceutical industry.
Biomarkers and Clinical Data to Optimize the Efficiency Of Therapies
According to the U.S. National Institute of Health (NIH) and the “Precision Medicine Initiative”, Precision Medicine is “an emerging approach for disease treatment and prevention that takes into account individual variability in genes, environment, and lifestyle for each person.”
Precision Medicine approach is the opposite of the “one-size-fits-all” approach, in which disease treatment or prevention strategies are driven with the model of an average person, with few consideration for individuals.
Precision medicine makes prevention and therapy more efficient or more adapted to each person in taking into account individual differences.
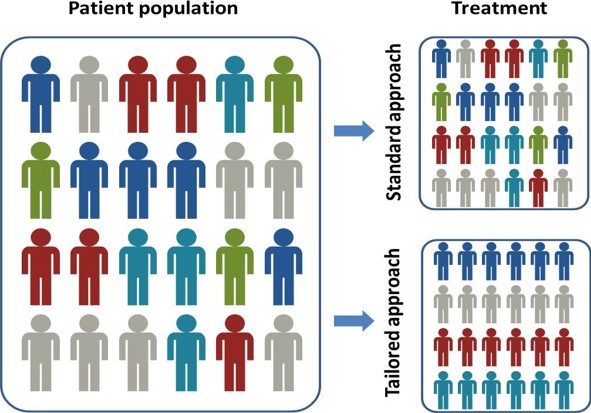
For instance, oncology is the medical domain in which Precision Medicine is already applied in routine. Sub-populations of patients are differentiated through the identification of molecular markers or molecular abnormalities. These molecular classifications allow to target therapies. Hence, physicians distinguished different types of cancer, associated with specific therapies.
It’s the case of breast cancer and trastuzumab treatment. This therapy is particularly efficient to block the overexpression of the HER2 protein (HER2+) and, consequently, the tumor growth. For instance, a recent clinical study showed that 89% of patients with early-stage HER2+ breast cancer and treated with adjuvant trastuzumab were disease-free survival at 4 years. In breast cancer treatment, other molecular diagnostics are also used in routine to determine if a patient is a “good responder” to chemotherapy or to immunotherapy.
But, as illustrated in these examples, implementing Precision Medicine in clinical settings requires the identification of specific sub-populations of patients. Each of them is characterized by a same or equivalent susceptibility to a disease development, a progressive behavior and/or a response to a specific therapy.
To achieve the identification of several sub-populations of patients suffered from the “same” disease needs to generate, integrate and analyze clinical, biological and/or physiological data from both healthy and disease states in individuals to characterize a patient’s disease progression or to measure the evolution/benefit of a therapy.
In practice, this approach needs and uses the identification and clinical validation of biological, physiological, or morphological markers. The challenge is to determine which marker or which combination of markers is reliable, relevant and specific to the measurement of a normal or pathological biological process, or to the measurement of the patient response to a predetermined treatment.
These biomarkers can be cellular or molecular (DNA, RNA, protein). They are measured from a tissue biopsy or a liquid biopsy (blood, urine…). Other biomarkers (morphological, etc.) can be also used or measured through radiological acts.
Data Driven Medicine
As already mentioned, implementing Precision Medicine in clinical settings requires to generate, integrate and analyze clinical and biological/physiological data to better understand diseases and develop and deliver more precise diagnostics, therapeutics, and prevention measures.

In other words, Precision Medicine can be considered as a data-driven approach applied to research and medicine with the objective to better follow people health and cure patients.
Precision Medicine or Data Driven Medicine will lead to the availability of new treatments, to a better use of innovative molecules and generic drugs, but also to generate cost savings in hospitalization, biomedical research and pharmaceutical industry.
Big Data and Artificial Intelligence (AI)
The integration of Data from clinics, phenotypes, genomics, transcriptomics, proteomics, etc. represents a huge and powerful instrument for physicians to obtain a more precise vision of diseases.
Nevertheless, the Big Data obtained from genomics, transcriptomics, proteomics, etc. needs today specific bioinformatics structure, software and algorithms due to their complexities and huge volumes.
For instance, the revolution of Next Generation Sequencing allows today to generate hundreds of millions of sequence data within one or few days when the Sanger method used in the beginning of the 2000’s could generated few dozens of thousands of sequence data.

Moreover, 80% of patient/clinical data is unstructured whereas clinicians need a global vision in order to make medical decision(s). To implement Precision Medicine in clinical settings, healthcare actors need tools and development to structure these Big Data and identify parameters/factors that could be situation predictive/clinical/medical behaviour negative for the patient. The combination of biological and clinical data, associated with public health data is a fundamental lever for Precision Medicine. However, major challenges such as the transfer of this data and the management of patients’ private data still require the development of specific tools and methods.
Several initiatives have emerged in the USA or in Europe to answer to the needs of the scientists to treat and analyze these new massive amounts of data, whatever the kind and the supplier of these data. For example, to face to this complex IT and big data challenge, several European organisms have been gathered in the PRACE initiative (Partnership for Advanced Computing in Europe, www.prace-ri.eu/) to provides access to several leading-edge high performance computing (HPC) systems (supercomputers) to researchers and scientists from academia and industry.
As already mentioned, biological data from genomics, transcriptomics, proteomics, etc. are crucial for predictive and precision medicine. Several approaches including: development of new biological/clinical databases development of algorithms and predictive models, the use of datamining platforms and Artificial Intelligence, became the essential supports to analyze data and to identify markers that will permit the development of innovative diagnostics or new therapeutics.
Hence, in a next future, Articificial Intelligence (AI) and machine learning will assist healthcare actors in their daily practice with more informed / precise clinical decision-making.

